Intro: In this lab, my partners, Ivy, Maia and I decided to try the naked egg experiment. Basically we put eggs summered in vinegar, for about a week. And over the course of the week we would check up on the egg and observe the process of the skin dissolving and the egg almost inflating. After a week has passed we would take out our egg and attempt to bounce them. After we experimented with all three of our eggs like bouncing them from different heights and observing them, we would go on online and research the science. To find out why the experiment turned out the way it did.
What happened: After a week our eggs were considerable lager maybe about one inch was added on in diameter. Also it smelled strongly of rotten eggs so we had to cover the beaker, although there was no sign of the egg rotting. After a day of the egg being in the vinegar the shell had already started to dissolve with bubbles surrounding the egg. After that you could easily see the egg breaking off and the reaming pieces of the shell floating on the top of the beaker. After the egg shell finished peeled off the egg began to grow more each day. By the 5th day though it appeared to have stopped growing but we still left it in the vinegar over the weekend so that it would continue to get a rubbery outside.The white of the egg was almost completely clear the yolk look the same. The egg felt much more dense then a hard boiled egg. After we observed our eggs one last time we were ready to test bouncing our eggs you could bounce it from a low spot perfectly. It was no bouncy ball but it defiantly was more bouncy then you would imagine a egg to be. If you actually bounced it with some force the outer shell would pop letting out all of the inside of the egg with the yolk still intact. All three of our eggs responded to bouncing the same.
On a Molecular Level: The vinegar easily removes the egg shells because vinegar contains an acid called acetic acid CH3COOH, which reacts with the high calcium content breaking up the carbon into the smallest terms of carbonate. In the egg causing it to dissolve and peel off. White vinegar usually contains 5% percent pure acetic acid and 95% water. As the eggshell dissolves, carbon dioxide is produced which we saw in the form of bubbles. Leaving the egg immersed in the vinegar for almost a week will cause its shell to dissolve completely. he explanation for the eggs bounciness comes from something called osmosis. Osmosis is the movement of liquid from a solution of lower concentration to a more concentrated solution through a slightly permeable substance. Permeable means fluids or gases are able to pass through the material. In the bouncy egg experiment, the vinegar slowly 'passes' through the eggshell until it dissolves it completely leaving the egg rubbery or leathery in texture. That texture is what makes it bouncy. Unfortunately the bouncy outer layer can not completely pass through the whole egg that is why it will pop when bounced from a high level. Jennessa told me the an egg is a single cell with the shell being the membrane of the cell and the white of the egg representing the plasma membrane which contains glycoprotein and lastly the yolk represents which represents the nucleus.
Take Aways:
Research:
http://www.factmonster.com/cig/science-fair-projects/making-eggs-that-bounce.html
https://explorable.com/magic-egg-experiment
http://www.ncbi.nlm.nih.gov/books/NBK26842/
nglerscience.com/lab/experiments/naked-egg-experiment
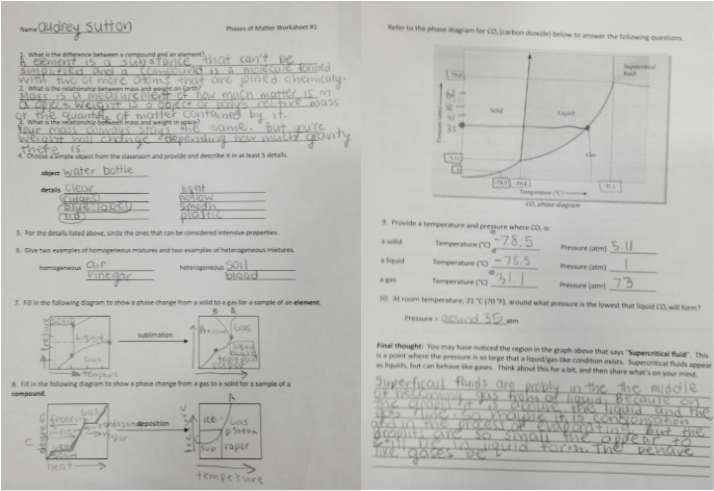
Reflection:
As far as how successful the product of lab turned out, I would say good because it turned out the way it should it was a very planed experiment without much room to mess up. I liked working with Ivy, and Maia they are both really reliable people. Patience was defiantly key in this lab if we had not waited a full week I don't think they would bounce as well. I wouldn't want to continue this experiment just because it is so simple. Except for I would like to know what would happen if we left the egg in the vinegar for even longer if the osmosis could pass all throughout the whole egg. Overall I think this experiment worked out really well and learned some new chemistry terms and topics that I have never hear of before.
Photos of the Process:
What happened: After a week our eggs were considerable lager maybe about one inch was added on in diameter. Also it smelled strongly of rotten eggs so we had to cover the beaker, although there was no sign of the egg rotting. After a day of the egg being in the vinegar the shell had already started to dissolve with bubbles surrounding the egg. After that you could easily see the egg breaking off and the reaming pieces of the shell floating on the top of the beaker. After the egg shell finished peeled off the egg began to grow more each day. By the 5th day though it appeared to have stopped growing but we still left it in the vinegar over the weekend so that it would continue to get a rubbery outside.The white of the egg was almost completely clear the yolk look the same. The egg felt much more dense then a hard boiled egg. After we observed our eggs one last time we were ready to test bouncing our eggs you could bounce it from a low spot perfectly. It was no bouncy ball but it defiantly was more bouncy then you would imagine a egg to be. If you actually bounced it with some force the outer shell would pop letting out all of the inside of the egg with the yolk still intact. All three of our eggs responded to bouncing the same.
On a Molecular Level: The vinegar easily removes the egg shells because vinegar contains an acid called acetic acid CH3COOH, which reacts with the high calcium content breaking up the carbon into the smallest terms of carbonate. In the egg causing it to dissolve and peel off. White vinegar usually contains 5% percent pure acetic acid and 95% water. As the eggshell dissolves, carbon dioxide is produced which we saw in the form of bubbles. Leaving the egg immersed in the vinegar for almost a week will cause its shell to dissolve completely. he explanation for the eggs bounciness comes from something called osmosis. Osmosis is the movement of liquid from a solution of lower concentration to a more concentrated solution through a slightly permeable substance. Permeable means fluids or gases are able to pass through the material. In the bouncy egg experiment, the vinegar slowly 'passes' through the eggshell until it dissolves it completely leaving the egg rubbery or leathery in texture. That texture is what makes it bouncy. Unfortunately the bouncy outer layer can not completely pass through the whole egg that is why it will pop when bounced from a high level. Jennessa told me the an egg is a single cell with the shell being the membrane of the cell and the white of the egg representing the plasma membrane which contains glycoprotein and lastly the yolk represents which represents the nucleus.
Take Aways:
- Osmosis
- Solution
- The chemical structure of vinegar
- The main sections of a animal cell
- The reactions that take place in the bouncy egg experiment
- Acetic acid
Research:
http://www.factmonster.com/cig/science-fair-projects/making-eggs-that-bounce.html
https://explorable.com/magic-egg-experiment
http://www.ncbi.nlm.nih.gov/books/NBK26842/
nglerscience.com/lab/experiments/naked-egg-experiment
Reflection:
As far as how successful the product of lab turned out, I would say good because it turned out the way it should it was a very planed experiment without much room to mess up. I liked working with Ivy, and Maia they are both really reliable people. Patience was defiantly key in this lab if we had not waited a full week I don't think they would bounce as well. I wouldn't want to continue this experiment just because it is so simple. Except for I would like to know what would happen if we left the egg in the vinegar for even longer if the osmosis could pass all throughout the whole egg. Overall I think this experiment worked out really well and learned some new chemistry terms and topics that I have never hear of before.
Photos of the Process:













 RSS Feed
RSS Feed