Intro: This was a class experiment where we got in table groups. And learned how to make simple batteries out of pennies and vinegar soaked cardboard and why they worked. We could use the battery we made to light a LED. Some of the chemical concepts involved with this experiment are; chemical RXNs involving energy, the flow of electrons, atoms, element attractions (negative and positive), the transfer of elements, cell volts, and electron solutions. I will explain how each of these concepts are evolved with this lab.
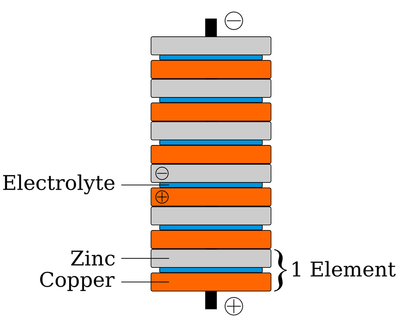
What happened: To start making the battery we took sevenpennies that are newer then 1982 because they are made of almost 98% percent zinc which is essential to make this battery. To expose the zinc we sanded it down on cement until it was a smooth silver surface. When we finished that we got seven square pieces of card stock soaked in vinegar and we staked all of that in order with the zinc facing upwards. It is shown in the digram bellow. We put that stack on top of a piece of tinfoil.
On a Molecular Level: This penny battery is a voltaic pile which uses pennies which act as the metal discs like on traditional voltaic pile. The pennies are stacked with pieces of electrolyte filled paper in this case vinegar. A electron solution in simple terms is a type of acid that conducts RXNS. Each cell of the penny can produce up to 0.8 volts and can be staked together to make even more volts from each cell. Because this battery is a 'wet cell' the effectiveness could be lessened as the electrolytes evaporate. Batteries convert the chemical RXNS energy of the two metals, interacting with the acid on the tin foil into electrical energy. Each cell is defined as a stack of a zinc penny, tin foil and a copper/zinc penny. Indicating that they could power an LED light, needing 1.7 volts, only three cells need to be used. As time goes on the amount of energy that the battery can provide decreases. A five cell penny battery can last up to 6 1/2 hours providing minimal voltage. The stack of cells is also known as a voltaic pile. The vinegar soaked paper between the battery cells is the (-) nickel source terminal connected to a (+) connected to the coper side of the penny. By leaving the electrolyte soaked paper between each individual battery cell, the electrolytes force copper and zinc ions to flow.
Take Aways/Important Notes:
Research:
http://en.wikipedia.org/wiki/Penny_battery
http://www.how-things-work-science-projects.com/coin-battery.html
Reflection: From this lab I learned the basics of a battery, like the main components of a battery are; negative and positive sides, electrolytes, copper, and iron. I always throughout that batteries were complicated, but from this lab I see that they really aren't. It is really cool to think about electricity and how we can gain energy from other places besides the typical places you would think energy is derived from. It gives me more hope that we will find alternative energy sources. Looking outward, I'm glad we did this lab in our class because it is something that I might not do on my own. I think that I could continue with another lab like this because I have heard that you can make batteries out of many things because many things have energy stored chemically that you could use. Our battery worked really well it worked, because we followed instructions and were sure to have our pennies zinc all the way showing and made sure that the card stock was filled with electrolytes. It was so exiting to see the LED light up! Looking inward, It was interesting working with a bigger group because it is harder to compromise but things get done faster. I may want to work with some of these people individually in the near future. I learned a lot in this lab and a may want to labs similar to this and experiment with making batteries out of different things.
What happened: To start making the battery we took sevenpennies that are newer then 1982 because they are made of almost 98% percent zinc which is essential to make this battery. To expose the zinc we sanded it down on cement until it was a smooth silver surface. When we finished that we got seven square pieces of card stock soaked in vinegar and we staked all of that in order with the zinc facing upwards. It is shown in the digram bellow. We put that stack on top of a piece of tinfoil.
On a Molecular Level: This penny battery is a voltaic pile which uses pennies which act as the metal discs like on traditional voltaic pile. The pennies are stacked with pieces of electrolyte filled paper in this case vinegar. A electron solution in simple terms is a type of acid that conducts RXNS. Each cell of the penny can produce up to 0.8 volts and can be staked together to make even more volts from each cell. Because this battery is a 'wet cell' the effectiveness could be lessened as the electrolytes evaporate. Batteries convert the chemical RXNS energy of the two metals, interacting with the acid on the tin foil into electrical energy. Each cell is defined as a stack of a zinc penny, tin foil and a copper/zinc penny. Indicating that they could power an LED light, needing 1.7 volts, only three cells need to be used. As time goes on the amount of energy that the battery can provide decreases. A five cell penny battery can last up to 6 1/2 hours providing minimal voltage. The stack of cells is also known as a voltaic pile. The vinegar soaked paper between the battery cells is the (-) nickel source terminal connected to a (+) connected to the coper side of the penny. By leaving the electrolyte soaked paper between each individual battery cell, the electrolytes force copper and zinc ions to flow.
Take Aways/Important Notes:
- All energy on earth is stored chemically
- All elements have different attractions to electrons
- When an atom from a different element come into contact with another the elements will transfer
- The flow of electrons makes electrons
- In the penny battery the electrons will flow from the zinc, through the vinegar cardboard to the copper
- Repeated stacking of cells will increase volts
- At contact with the electron solution, each side of the penny forms positive ions (Zn2+) and (N2+)
- Zn losses electrons more easily so they travel to the Cu2+ ions. (Zn is oxidized) is oxidized (Cu+2) is reduced
Research:
http://en.wikipedia.org/wiki/Penny_battery
http://www.how-things-work-science-projects.com/coin-battery.html
Reflection: From this lab I learned the basics of a battery, like the main components of a battery are; negative and positive sides, electrolytes, copper, and iron. I always throughout that batteries were complicated, but from this lab I see that they really aren't. It is really cool to think about electricity and how we can gain energy from other places besides the typical places you would think energy is derived from. It gives me more hope that we will find alternative energy sources. Looking outward, I'm glad we did this lab in our class because it is something that I might not do on my own. I think that I could continue with another lab like this because I have heard that you can make batteries out of many things because many things have energy stored chemically that you could use. Our battery worked really well it worked, because we followed instructions and were sure to have our pennies zinc all the way showing and made sure that the card stock was filled with electrolytes. It was so exiting to see the LED light up! Looking inward, It was interesting working with a bigger group because it is harder to compromise but things get done faster. I may want to work with some of these people individually in the near future. I learned a lot in this lab and a may want to labs similar to this and experiment with making batteries out of different things.





 RSS Feed
RSS Feed